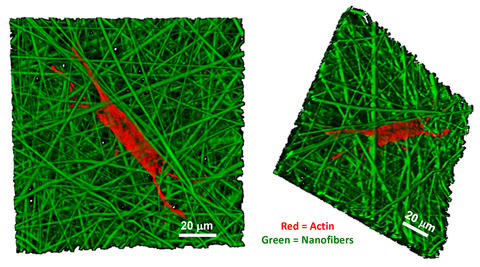
Bone-like cell growth on nanofibers: confocal microscope images detail the growth of a human bone marrow stromal cell (actin filaments in the cell "skeleton" are stained orange) on a nanofiber scaffold (green). The structure of thin fibers encourages stem cells to develop into the elongated, branched form characteristic of mature bone cells.
"Form follows function!" was the credo of early 20th century architects making design choices based on the intended use of the structure. Cell biologists may be turning that on its head. New research* by a team working at the National Institute of Standards and Technology (NIST) reinforces the idea that stem cells can be induced to develop into specific types of cells solely by controlling their shape. The results may be important to the design of materials to induce the regeneration of lost or damaged tissues in the body.
Tissue engineering seeks to repair or re-grow damaged body tissues, often using some form of stem cells. Stem cells are basic repair units in the body that have the ability to develop into any of several different forms. The NIST experiments looked at primary human bone marrow stromal cells, adult stem cells that can be isolated from bone marrow and can "differentiate" into bone, fat or cartilage cells, depending.
"Depending on what?" is one of the key questions in tissue engineering. How do you ensure that the stem cells turn into the type you need? Chemical cues have been known to work in cases where researchers have identified the proper additives—a hormone in the case of bone cells. Other research has suggested that cell differentiation on flat surfaces can be controlled by patterning the surface to restrict the locations where growing cells can attach themselves.
The experiments at NIST are believed to be the first head-to-head comparison of five popular tissue scaffold designs to examine the effect of architecture alone on bone marrow cells without adding any biochemical supplements other than cell growth medium. The scaffolds, made of a biocompatible polymer, are meant to provide a temporary implant that gives cells a firm structure on which to grow and ultimately rebuild tissue. The experiment included structures made by leaching and foaming processes (resulting in microscopic structures looking like clumps of insect-eaten lettuce), freeform fabrication (like microscopic rods stacked in a crisscross pattern) and electrospun nanofibers (a random nest of thin fibers). Bone marrow stromal cells were cultured on each, then analyzed to see which were most effective at creating deposits of calcium—a telltale of bone cell activity. Microarray analysis also was used to determine patterns of gene expression for the cultured cells.
The results show that the stem cells will differentiate quite efficiently on the nanofiber scaffolds—even without any hormone additives—but not so on the other architectures. The distinction, says NIST biologist Carl Simon, Jr., seems to be shape. Mature bone cells are characteristically long and stringy with several extended branches. Of the five different scaffolds, only the nanofiber one, in effect, forces the cells to a similar shape, long and branched, as they try to find anchor points. Being in the shape of a bone cell seems to induce the cells to activate the genes that ultimately produce bone tissue.
"This suggests that a good strategy to design future scaffolds would be to take into account what shape you want to put the cells in," says Simon, adding, "That's kind of a tall order though, you'd have to understand a lot of stuff: how cell morphology influences cell behavior, and then how the three-dimensional structure can be used to control it." Despite the research still to be done on this method, the ability to physically direct cell differentiation by shape alone potentially would be simpler, cheaper and possibly safer than using biochemical supplements, he says.
The work was supported in part by the National Institute of Dental and Craniofacial Research, National Institutes of Health.

