PML scientist Zeeshan Ahmed lowers a sample into the instrument chamber for analysis.
In living organisms, biomolecules such as proteins are constantly in complex motion, bending and flexing in different ways at different points. Each molecule has its own vibrational dynamics which affects its propensity to bind to adjacent molecules, such as pharmaceuticals. The number and nature of a biomolecule's vibrational modes depends on numerous factors including conformational variety, hydration state, solvate environment, peptide sequence, crystalline order, and more.
Characterizing and understanding the functional effects of those properties is the goal of terahertz (THz) spectroscopy, a newly emergent field that employs frequencies between microwaves and infrared radiation to investigate the structure and behavior of biosystems by examining their vibrational response. Academic labs, however, generally use low resolution spectrometers and often lack expertise in computational chemistry which limits the usefulness of their research.
Researchers from PML's Sensor Science and Radiation and Biomolecular Physics divisions are taking a leading role in the worldwide effort, providing critically important baseline data with exquisitely sensitive high resolution apparatus, painstaking methodology, and attention to the often dramatic differences between theory and observation.

"The sources and detectors for this work simply did not exist until very recently," says Zeeshan Ahmed of the Temperature & Humidity Group. "In fact, this frequency range used to be called the 'terahertz gap.' But advances in telecommunications technology have now made it possible for us to explore previously elusive aspects of biomolecules."
"Of course, there are many other sources of information. But they don't yield the insights we're looking for. X-ray crystallography, for example, will give you a nice, sharp picture of a single conformation, you know exactly where all the atoms are. But in your body, where proteins actually matter, they're constantly moving."
Similarly, says David Plusquellic of the Biophysics Group, "you can learn a lot from infrared (IR) spectroscopy. But the vibrational modes that show up in IR are usually localized within specific regions of the molecule. The THz modes, by contrast, involve the collective motions of all the atoms in a molecular structure."
In THz spectroscopy, researchers scan an isolated sample with a range of frequencies, typically a subset of the spectral region from 0.06 THz to 10 THz. Each specific frequency that is resonant with one of the molecule's multiple vibrational modes will be absorbed; the rest will pass through the sample to a detector. The resulting data yield absorption spectra that can reveal subtle but important differences between molecules that appear virtually identical in IR.

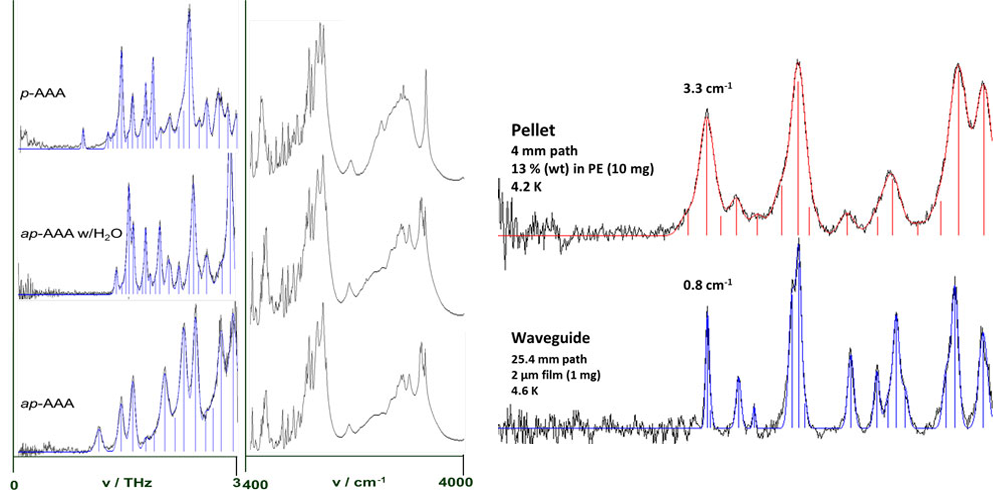
At PML, THz measurements are conducted in a multi-stage process designed to maximize the amount of information about the target molecule. The investigators carefully study x-ray and IR spectra of the molecule, and also calculate the vibrations predicted by quantum chemistry theory. Then a sample is scanned in vacuum at room temperature, at 77 K, and at 4.8 K. The coldest temperature generates the sharpest absorption features.
The sample can be combined with a filler such as polyethylene, which is transparent in the THz regime, and compressed into a disk about 1 cm wide and 1 mm to 3 mm deep. (See figure 3.) Alternatively, the sample can be measured in a waveguide formed by two gold-coated plates about 250 µm apart with the thin-film sample in between. This technique produces spectra with much higher line resolution than the pellet data.
Among other uses, THz spectroscopy holds considerable promise for studying how the motions of biomolecules change when they are bound in various ways to water molecules, which affects the molecules' conformation and function. Ahmed, Plusquellic, and colleagues recently conducted a study that demonstrated how the presence of water changes the structure and vibrational modes in three different peptides common to protein systems, and found that THz spectroscopy can very clearly distinguish those variations. The authors also determined the energetically preferred states of crystal hydration and suggested ways of more closely reconciling theoretical calculations with observed spectra. The extreme sensitivity of THz to solid forms of materials and the presence or absence of water in these materials has obvious applications in process engineering, crystal engineering, IP patrolling, quality control and identification of counterfeit drugs.
In PML, this methodology has recently been extended to pharmaceuticals. "Detecting the presence, nature, and effects of hydration can have large practical significance for quality control of pharmaceuticals," Ahmed says. "In general, dehydrated drugs are absorbed faster in the body, so the hydration state can change patient's drug exposure. This obviously impacts the drug's dosing profile approved by the FDA. The solid form has to be the same not only over the entire production campaign spanning many years but also during storage leading up to consumption."
"Results of this sort are very encouraging," says Plusquellic. "But there are major challenges that we will have to overcome if THz spectroscopy is to achieve its full promise. For example, there are many instances in which we are not sure what the spectra are telling us. And very often we find that the theoretical calculations are completely out of agreement with our observations. Indeed, one of our most important contributions to the field is providing sets of experimental data against which the theory can be tested."
The researchers are gradually working their way up to progressively more complicated samples. "For a long time, we concentrated on peptide chains," Ahmed says. "But over the last couple of months, we have switched to doing more pharmaceuticals. We now have data for non-steroidal anti-inflammatory drugs like Naproxen and Norfloxacin and antibiotics like Amoxicillin and Ampicillin.
"One day we'd like to be able to characterize a complex made up of a drug bound to a peptide or protein. X-ray crystallography doesn't reveal hydrogen bonding interactions, but THz vibrations do. And knowing the placement and behavior of hydrogen bonds can go a long way toward answering some very fundamental questions, such as how the complex stretches and contorts itself, exactly what happens when the molecules bind, and what constitutes a 'good fit.'
"In the long run, we want to help theory to become better, so that many steps in drug discovery can be done by computer. That could reduce research costs dramatically and help the pharmaceutical industry bend the 'Eroom's Law' curve, which is the decline in new drugs approved per unit of development cost, despite major technological advances, R&D support, and changes in regulatory climate."
Many observers believe that the steep rise in the cost of new drug development has negative consequences for nation's economy, budgetary outlook, and delivery of healthcare to a rapidly aging population. In those circumstances, in silico screening of drug candidates may be an important tool because it provides a cost-efficient way to design and develop drug candidates. The THz work at the PML is focused on providing baseline benchmarks that can be used to improve the accuracy of these computational methods, and to help researchers select a computational method best suited for their particular need.
Photographs by Aakash Patel