Spectroscopy: A Measurement Powerhouse

Spectroscopy. It may not be a household term, but it is one of the most popular and powerful measurement techniques in science. Scientists use spectroscopy to analyze starlight and other signals from outer space, to define the ticks in atomic clocks, to detect chemical pollutants in the air, to determine the composition of soil, clothing, trash and more, and to sniff out markers of disease and drugs in people’s breath. Recent advances in physics and measurement science have made spectroscopy more important than ever.
Given how crucial spectroscopy is to science — especially the measurement science NIST specializes in — we should probably understand how it works. Here are the basics.
What is spectroscopy?
Spectroscopy uses the interaction between light and matter to gather information. So to understand spectroscopy, we first need to answer a more basic question: What is light?
Light is one of the main ways that nature sends energy from place to place. Light travels in the form of electromagnetic waves that rise and fall as they speed through space, like the alternating crests and troughs of water waves traveling across the ocean. The number of times a light wave rises and falls in one second is known as its frequency.
Light waves — also known as electromagnetic radiation — range from extremely-low-frequency electromagnetic waves that cycle just a few times per second to ultra-energetic gamma rays from space cycling more than 10 to the 28th power (10,000,000,000,000,000,000,000,000,000, or 10 trillion quadrillion) times per second!
The fact that light comes in so many different frequencies is crucial to our ability to sense our world. Indeed, the human eye is a type of spectrometer. It can distinguish millions of colors — each revealing a light wave of a slightly different frequency.
Yet visible light makes up only a tiny part of the overall electromagnetic spectrum.
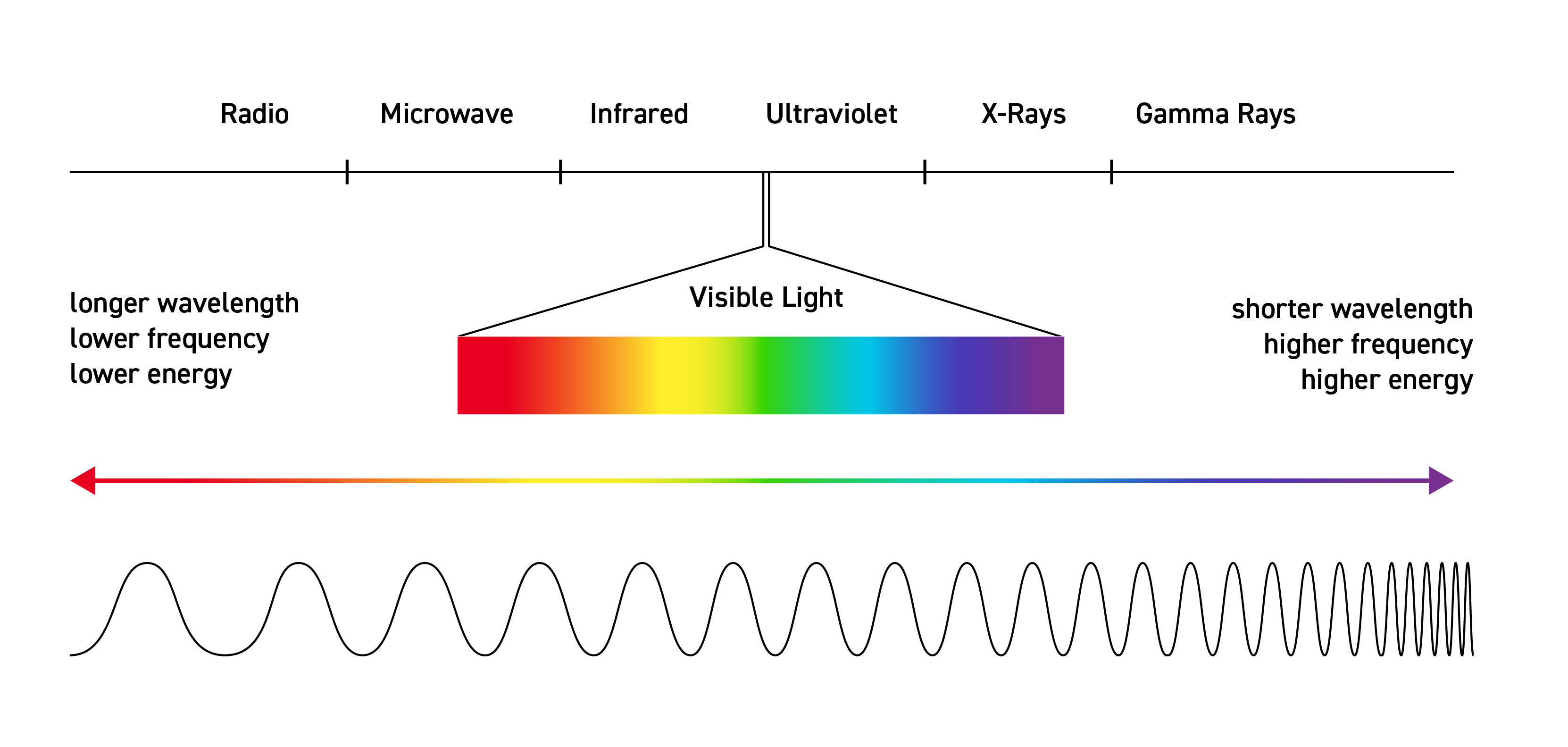
Spectroscopy uses the full spectrum and the fact that all atoms, molecules and objects — from tiny molecules to massive stars and planets — absorb and radiate light. The specific colors, or frequencies, that different gases and objects emit or absorb can tell us about their identity, composition, concentration and temperature.
For example, stars, with broiling-hot surface temperatures of thousands of degrees, radiate mostly visible light, as well as ultraviolet and infrared frequencies.
By contrast, objects around room temperature — including our bodies — radiate mostly in the infrared, a range of light with frequencies below that of visible light. Because many common molecules absorb and emit infrared light, infrared spectroscopy is extremely important in fields such as chemistry, atmospheric science and materials science.
Meanwhile, many objects in space radiate in lower- and higher-frequency parts of the spectrum such as radio waves, X-rays and gamma rays, making these frequencies important in astronomy.
How does spectroscopy work?
The key to spectroscopy is having a way to precisely measure light frequencies. From early studies of light going through prisms, scientists have invented many kinds of spectrometers to separate light into individual colors and determine their frequencies. Examples include Fourier-transform spectrometers and optical frequency combs.
Scientists have developed many different kinds of spectroscopy for a wide variety of purposes. For the rest of this article, we will focus on two major types.
In emission spectroscopy, scientists measure light frequencies emitted by an object like a star. For example, to discover exoplanets, scientists look for tiny changes in the spectra of distant stars compared to the unchanging spectrum emitted by special lamps.

Another growing application area is laser-induced breakdown spectroscopy. A laser pulse turns a small amount of a sample into a hot plasma, which emits light as it cools. Scientists analyze the spectrum of the emitted light to determine the makeup of the sample.
Scientists and engineers use emission spectroscopy to:
- Design lighting
- Plasma-etch computer chips
- Analyze plasmas created during nuclear fusion
- Study the origins of heavy elements
- Analyze Martian rocks and Earth rocks
- Study the Sun
- Identify healthy and diseased crops
- And do many other things!
In absorption spectroscopy, scientists measure light that has passed through clouds of atoms or molecules.
Each type of atom or molecule absorbs a unique set of light frequencies, creating what’s known as a “spectral fingerprint.” Just as your fingerprint can identify you among the billions of people on Earth, the absorption spectra of molecules can be used to identify them.

The spectral fingerprint of an atom or molecule depends on the internal configuration of its electrons, protons and neutrons. When light of the right frequency is absorbed by an atom or molecule, its energy causes electrons to rearrange themselves into a higher-energy configuration.
Scientists therefore look for missing frequencies of light that are known to be absorbed by atoms or molecules of interest. By precisely measuring how much light is missing at various frequencies, scientists can determine whether the atom or molecule is present in a sample, as well as how much of the atom or molecule there is.
Absorption spectroscopy helps scientists do many things, including:
- Sort textiles and clothing for recycling
- Detect markers of Covid-19 and other pathogens in exhaled breath
- Detect cancer
- Analyze blood
- Identify complex proteins
- Count microplastics in the environment
- Study the atmospheres of faraway exoplanets
- Measure the concentrations of greenhouse gases in the atmosphere
You may not think about spectroscopy on a daily basis, but behind the scenes, this powerful and versatile measurement tool impacts your daily life.
Learn More
Shedding Light: How NIST’s Light Database Helps Bring You Computer Chips, Welded Steel and More: This blog post describes the many uses of NIST’s atomic spectra database.

