Redefining the Mole
Spectrometers, Silicon Spheres and Statecraft Modernize Chemistry’s Mammoth Measurement Unit
In suburban Maryland, on the third floor of the Advanced Chemical Sciences Laboratory at the National Institute of Standards and Technology (NIST), Bob Vocke and Savelas Rabb are explaining how they are helping to redefine the mole, that mammoth concept we learned in high-school science class.
Mostly, this means using abstract symbols and numbers, dancing along in a long equation that Vocke obviously thinks is beautiful. You can tell because he enthusiastically writes each detail out on the white board for some lab visitors. Every so often, Rabb provides suggestions or tells a joke and sort of eggs Vocke on. It is clear they are having a good time and could do this kind of annotation and explanation all day.
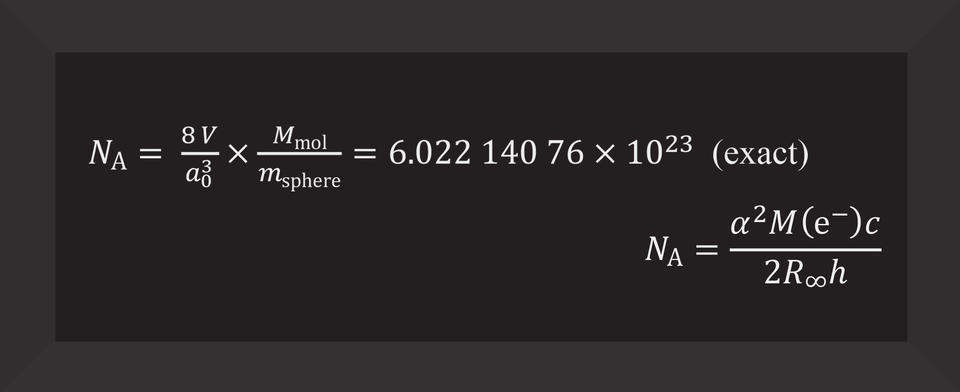
After these equations, they add more:

To fully grasp the new definition of the mole, you must embrace these long equations, which show how that measurement connects to fundamental constants of the universe—such as the speed of light in vacuum and the amount of charge in an electron. In turn, these fundamental constants have completely redefined the modern metric system, known as the International System of Units (SI). The mole is one of the seven base units of the SI.
For Vocke and Rabb, the equation work brings joy. The process is entertaining, and a little fun.
For most others, it seems impossibly cryptic.
On the surface, the mole’s basic definition remains the same as it was before. It’s a measure of stuff—how many molecules or atoms you have of a particular substance such as water, or gold or a protein.
But the earlier definition was more complicated than it needed to be. Previously, a mole was the amount of substance that contained as many elementary entities as there were atoms in 0.012 kilograms of the most common form of carbon, known as carbon-12. A further complication: The mole relied on another definition, the definition of the kilogram, which was then specified by the mass of a platinum-iridium cylinder locked up in a special vault outside Paris, France.
The work that Vocke and Rabb have done has helped to unlock that vault and free both the mole and the kilogram. But to understand how, one has to first embrace the complex equation Vocke’s written out.
Maybe not *understand* it. But embrace it.
Because what Vocke, Rabb and numerous other scientists have done over the course of many years and through many lab experiments using enriched isotopes, sophisticated instruments and loads of chemistry is explained right there, in the symbols and the numbers, clearly and concisely.
Both men pause from their mathematical annotations to say something reassuring: You need not be a math genius to grasp what’s happened. This is good news to a math-phobic visitor.
But to understand how the mole’s definition has changed, you do have to know a few details about the measurements, their importance to the world’s social and economic order, and their shorthand notations.
In practical terms, the mole helps chemists measure stuff. It helps express the amounts of atoms or molecules in a chemical reaction. Cause a half-mole of oxygen molecules (O2) to react with a mole of hydrogen molecules (H2) and you get a mole of water (H2O)—equal to about 18 grams of substance. That’s not very much water—it would create a pretty shallow pool in a small paper cup.
However, thanks to the concept of the mole, you know that 18 grams of water—a macro quantity you can easily see and measure—contains approximately 600 billion trillion individual water molecules—a submicroscopic quantity not easily measured. And you would know that for every 10 water molecules you create, you need exactly 10 oxygen atoms and 20 hydrogen atoms. Because of this, you can easily design chemical reactions at the atomic level that would produce exactly the amount of substance you need at a macroscopic level.
For about 20 years, the international community worked on the modern redefinition of the mole. This effort centered on an amazingly ultra-pure sphere of one of the world’s most common elements: silicon. But unlike the kilogram ingot in France, this one doesn't sit in a little vault somewhere to be used for reference. Instead, the equations themselves are the reference. The sphere serves as evidence that the equation works.
And scientists have finally achieved what French revolutionaries envisioned when they created the SI: a measurement system for all times and for all people.
Even describing the scientific work that Vocke and Rabb finished involving the sphere brings a contrarian smile to Vocke’s face. No one has to be beholden to a drifting artifact anymore. It is as if a king has been dethroned or a monarchy toppled, and it seems as if that appeals to the rebellious side of Vocke’s personality.
What Vocke wants, really, is for the public to grasp the importance of what has just happened. This isn’t about reputation or professional pride. This is about leveling the playing field. An important part of his vocation has been democratized, thanks to international cooperation between scientific teams from all over the globe. Now, no one needs to travel to France and get permission to compare their measurement of mass to a special kilogram artifact in a little vault.
With the sphere, anyone can figure out if their measurement of the mole matches those in Russia, Germany, Japan, South Africa or Nepal. Well, maybe not just anyone. But anyone who can comprehend this kind of complex formula. Anyone with a powerful analytical instrument known as a mass spectrometer. And a very pure silicon sphere. And a very powerful X-ray machine that can measure the spacing of atoms in the samples.
To a member of the public, this still seems akin to visiting elite scholars with hidden knowledge. These two scientists helping to redefine the mole, Vocke and Rabb, still seem like scientific illuminati.
Then they begin to talk about silicon, and the numbers melt away into something tangible, and it becomes easy to appreciate and share their rosy feelings.
This, then, is what you can focus on when you try to comprehend the amazing feat that scientists have just pulled off: the equation for the mole isn’t just a theorem, a mind exercise. It increases international access to one of the most important bridges connecting the atomic and the macro worlds. And silicon is the material we will use to build that bridge and traverse those realms with Vocke and Rabb.
Silicon Bridges
The word “silicon” has become a huge part of the modern American lexicon. Every day, people use the phrase Silicon Valley as shorthand for the entire world of modern IT and computing and digital innovation. As in: Let’s see what Silicon Valley has to say about that new policy. Others refer to silicon chips as a catch-all phrase for all computer parts.
But most people using such phrases would stop, dumbfounded, if you ask them for a definition.
What is “silicon”?

The word refers to an element of the periodic table, second only to oxygen in abundance on planet Earth. As an element, it is a significant building block for mineral compounds and a substance that exists in three different forms; silicon-30, silicon-29 and silicon-28. Take a hike in the woods and silicon in the form of silica is all around you in the soil, in the rocks, in the plants, because it links with oxygen to form silicates, the most common category of minerals that includes feldspar, quartz and mica. Silicon is also in beach sand, again present as silica, which is then melted down to make glass. There’s even silica in our bones; it’s essential for our body.
Rabb, however, struggles to find a sample of silicon to show a visitor.
“We don’t really use silicon that often in our labs,” he says with an apologetic grin.
Finally, he finds a chunk stored safely away in the back of a drawer. He pulls out a plastic baggie and there it is inside, sharp-edged and gleaming. He breaks the bag’s ziplocked seal and lays the piece across his palm for inspection.
When the NIST scientists received silicon from Germany’s national measurement institute as part of the effort to redefine the mole, it arrived as small bits, not unlike a collection of strange, small shiny ice-cream sprinkles, ready to be dissolved and eventually analyzed by the big instrument called a mass spectrometer that sits at the center of the lab. This random silicon piece in Rabb’s hand is some kind of novelty, but nonetheless, a piece of the stuff that can be handled, examined and held up to twinkle under the laboratory light fixtures. It is hard but incredibly light, like a party favor or a toy. Difficult to imagine that it provides the ammunition for a revolution in measurement science, or “metrology,” as the scientists call it (not to be confused with “meteorology”).
Although the sample Rabb just pulled from his laboratory stock looks metallic, silicon is not a metal but rather a metalloid. And although found nearly everywhere in nature as silica, you would have a tough time finding it in this metalloid form except around some very rare places, near active volcanoes or as inclusions in gold. That’s because it is almost always bound up with other elements. So even though it is part of the things around us, it can’t be mined in its silicon form. It must usually be isolated or purified and coaxed to form crystals useful for human endeavors.
The silicon components in computers—often called chips—are really wafers of silicon that are made by purifying, melting and cooling the crystals into an ingot shape that can be sliced into very thin pieces. The slices or wafers become “semiconductor chips” when other elements are added into them to create electronic circuits.
Elementary Molecules: Rock Me, Amedeo
Perhaps no one would be more fascinated by silicon and its role in the effort to redefine the International System of Units (often referred to by the French term Système International d’Unitès, or SI) than Amedeo Avogadro, for whom a constant of nature has been named.

Avogadro lived from 1776-1856. He began his career as a lawyer, later becoming a chemist when his curiosity about the natural world led him to interrogate it. He famously hypothesized that equal volumes of gases contain equal numbers of molecules if held at the same temperature and pressure. He also asserted that simple gases were made of compound molecules of multiple atoms. His work was so early in the game of chemistry that the concept of an “atom” was not even widely embraced at the time. He called them “elementary molecules.”
Avogadro may have been a great thinker, but he was not very good at convincing other people about his ideas. It took 50 years for others to catch up and realize he had been onto something big with all these small “elementary molecules” making up the world. By then, poor Avogadro had long gone to the grave.
Historians, mathematicians and chemists love to argue about why his concept was initially disregarded and took so long to catch on. In the end, what’s essential to know is that his fundamental information about gases would lead to a greater understanding of all substances.
To honor his discovery, it was decided early in the last century that the number of molecules in one gram-molecule of oxygen would be called the “Avogadro number.” With the introduction of the mole to the SI in 1971, this definition was changed to the “Avogadro constant.”
As noted before, a mole is the amount of substance that contains as many elementary entities as there are atoms in 0.012 kilograms of carbon-12. The Avogadro constant for carbon-12 is written out as: 6.022×1023, or 602,200,000,000,000,000,000,000. This huge number is usually referred to as NA.
Technically, Avogadro didn’t invent the mole—in fact the term didn’t appear until almost 1900, taken from the abbreviation Mol, from the German word for “molecule” (“Molekül”). But we can thank him for being an important part of the concept.
Playing the Mass Spec
Avogadro mostly worked with rudimentary instruments: small burners and such. Here in the NIST labs and pretty much everywhere modern chemists work, there are now instruments called mass spectrometers, which are often large boxy things. To Avogadro’s 19th-century eyes, these might have looked like child-sized coffins or large trunks for sending goods overseas via tall ships.
Mass spectrometers are used to vaporize samples into atoms and molecules and then ionize them (making them electrically charged). Inside the big box, those ions are accelerated by an electric field and then deflected by a magnetic field where they acquire a trajectory based on their mass- to-charge ratio. Lighter ions are more strongly deflected while heavier ones undergo less deflection. Researchers obtain a spectrum of masses in a sample by observing the different deflections. These mass spectra are like unique fingerprints that can help identify the composition of the original sample. Natural silicon, for example, has three stable isotopic forms and produces a mass spectrum consisting of silicon-28, silicon-29 and silicon-30.

It is easy to get the sense that Rabb really loves to use his mass spectrometer the way someone else might like playing a musical instrument or writing tight and concise code for computers. He likes thinking through the process. He tries to understand how each step will influence the precision of the measurements that result from his work.
Growing up, Rabb had two equal interests in school: history and chemistry. His friends were shocked when he listened to the counsel of his mother, who told him that science would make a more lucrative career choice. His friends thought he was going to be a teacher. To him, however, history and chemistry were not all that different. Both, he says, involve understanding contexts and questioning the world’s assumptions. Both feed his intense and insatiable love of factual knowledge. He’s worked at NIST for 15 years, often using different types of spectrometers to examine things as varied as soils, sediment, cement, solder, blood and coal.
Vocke and Rabb are a study in contrasts. Vocke is animated and loves to popcorn from one interesting topic to another in conversation. He speaks in short but fluid anecdotes and sits in an office filled with books about hiking through Utah and big hunks of rock he’s collected while travelling mountain ranges all over the world. He began his career in the late 1960s as an engineer, then switched to geology and isotope geochemistry before finally settling into a research position at NIST in the mass spectrometry group. In addition to English, he speaks German and French. He always answers the phone with two sharp syllables: Voc-ke (pronounced to rhyme with the phrase “okey-dokey”).
Rabb is careful, quiet and contemplative, and speaks softly. When listening to someone else speak about a demanding subject, he comes to a standstill and his body adjusts to full receiving mode. He likes precision. And to him, there’s always a need to be exacting and careful in how you set up an experiment.
Although extremely serious about science, they both laugh a lot, often at each other’s jokes.

“It’s easy to work with Bob,” Rabb says. “He is so open to new ideas and talking through the problems. He was always inviting me to make suggestions and was amenable to new ideas.”
Vocke, on the other hand, credits Rabb’s chemistry skills.
“He is, in my opinion, an exceptional chemist,” says Vocke of his collaborator. “He understands, maybe he even feels the atoms … he is just very quiet, precise, exacting.”
They were brought together in 2010 to work on the molar mass measurement challenge of the Avogadro project, and the resulting collaborative work would relate to the long equation that Vocke was so excited about.
“Our part was a small part,” Vocke says. “But a critical part. Really, this was an international effort at its best.”
Measured Diplomacy
The redefinition of the mole has been intertwined with that of the kilogram throughout history. And the kilogram has always taken center stage when it comes to the SI redefinition. As the unit of mass, the kilogram was the last unit based on a physical artifact—a cylinder made of platinum-iridium stored at the BIPM (International Bureau of Weights and Measures, or Bureau International des Poids et Mesures) headquarters in a suburb just outside of Paris, France. Other SI base units tied to the kilogram included the mole, the ampere (used to measure electrical current) and the candela (which measures the luminous intensity of light) as well as many SI-derived units such as the newton (to measure force), pascal (to measure pressure), joule (to measure energy) and watt (to measure power).
Both the metric system and efforts to establish and agree on a definition for worldwide measurements date back to 1875, when representatives of 17 nations (including the U.S.) signed a treaty known as the Meter Convention in Paris. This treaty also established a group of international organizations devoted to measurement. The treaty was modified slightly in 1921 but remains the basis for all international agreements on the seven base SI units of measurement like the mole, the meter and the kilogram.
The international prototype of the kilogram (often referred to as the IPK) was sanctioned as the unit of mass in 1889 at the first gathering of a group called the General Conference on Weights and Measures (CGPM, Conférence Générale des Poids et Mesures). In 1960, the SI was established by the 11th meeting of this international organization. The CGPM still exists and consists of delegates from around the world who meet every four years to ensure the accurate dissemination of the SI through national metrology institutes such as NIST. The group also modifies the SI as needed to reflect the latest advances in science and technology.
All these acronyms can be overwhelming, and it can feel like swimming in alphabet soup.
But here’s what’s important to know. Previous to 1889, a kilogram was the mass of a cubic decimeter of water. But that year, the CGPM sanctioned the new international prototype—the IPK—at its first meeting. The IPK was an actual object that sat like a crown jewel, locked securely away in a little vault. Only certain people could handle it, and they had to use special instruments and take extreme care as they did so.

“Think about that,” said Willie May, a former NIST director who is now vice president of the International Committee for Weights and Measures (CIPM, Comité International des Poids et Mesures). “Suppose you dropped [the IPK] and chipped a little bit off. Well, the kilogram is defined as the mass of the IPK. Technically, all the mass measurements in the world would be offset by that amount.”
This notion itself sounds like a plot from an international thriller in which the villain could plunge the world into chaos by knocking a small bit off the IPK. The world’s measurement vulnerabilities appeared to be quite out of step with modern times and global trade and commerce. The little artifact in the vault seemed like a throwback to an era of emperors.
The CIPM oversees the BIPM, which has the task of ensuring worldwide harmonization of physical measurements.
The CIPM recommends any needed modifications to the CGPM for formal adoption. The CIPM may also, on its own authority, pass clarifying resolutions and recommendations regarding the SI (these resolutions and recommendations usually deal with matters of interpretation and usage).
Although it may feel a bit excessive when you first contemplate all the diplomacy described in this process, measurements are really agreements between humans. Disagree by even one drop in your measurement and the price of a barrel of something could be mispriced by an enormous amount of money. Or consider the dosage of medicines. The doctor says one thing, but your measurement at home when you take the prescription is different. The results could be deadly. Measurements matter, and our ability to take them for granted is, in a way, a hallmark of prosperity. Agreement is akin to peaceful commerce.
As the national metrology institute for the U.S., NIST has been working on better ways to define the kilogram for a long time. Reassuringly, a lot of what the CIPM does sounds more like a science fair than an adventure movie, especially the way May explains it. There is no evil force in the world trying to take down the world’s measurements by destroying the IPK artifact. There’s just a desire to make measurement more exact and accessible.
Constantly Walking the Planck
The road to democratizing measurement involves a quantity from quantum physics called the Planck constant. Conceived by physicist Max Planck in 1900, this constant was introduced at the dawn of quantum mechanics, the theory that describes the behavior of atoms and other things at atomic and subatomic scales too small for us to see.
While the value of the mole is almost unimaginably big, Planck constant is a tiny number, containing 33 zeroes after the decimal point. According to quantum mechanics, an atom only absorbs or doles out energy in package-like chunks or “quanta,” whose value is equal to a multiple of Planck’s constant. It’s like a monetary system in which pennies don’t exist; you can only buy or sell things in multiples of nickels. The nickel, in this case, is Planck’s constant.
How does this relate to mass? Well, this brings in the most famous equation in all of physics: E=mc2. This says that mass can transform into energy, and energy into mass. And one type of energy is equivalent to another type. So, electrical energy can transform completely into mechanical energy, and vice versa. If the electrical energy of a quantum system is proportional to Planck’s constant, and it creates a force that balances out the weight of a mechanical system containing a mass, you can measure the amount of that mass using Planck’s constant.

Unlike IPK in the little vault outside Paris, Planck’s constant is never going to change because it is a fixed constant of nature, May says. By redefining the kilogram using an accurate value of that constant, we have something fixed and always available. “So, we need to determine what that number truly is, and then move forward with that equation.”
The different nations around the world were asked to put their best efforts forward to answer the question: What is the most exact way to measure the Planck constant? And, how will we know that we are all measuring the exact same thing without the use of an actual thing.
Scientists have come up with two ways for accurately measuring the Planck constant. One involves using an apparatus called a Kibble balance, which measures how much electrical power is needed to support a kilogram mass. This approach uses electricity and quantum phenomena. In other words, physics.
The other, a more indirect approach, uses X-rays focused on a silicon sphere to count the number of atoms in a mole of silicon. This has been done on two highly purified and polished crystal silicon spheres named S5 and S8.
This approach does double duty. It provides a measurement of the Avogadro constant—for a modern definition of the mole. And since Vocke’s beloved equations link the Avogadro constant to the Planck constant, the measurement enables researchers to calculate Planck’s constant to arrive at a modern-day definition of the kilogram.
This second approach does not depend on quantum mechanics and can be seen as a counting exercise, very complicated and quite sophisticated, yes, but in the end just a counting exercise. Scientists need to be sure that everyone is measuring the same amount of silicon in a similar manner, using the same processes so that the numbers are consistent and comparable. And we need to know that the measurements are made using the purest material possible.
In other words, chemistry.
It is at this point in the explanations that Vocke gets asked if the mole in this context is theoretical, an idea, a notion that is more philosophical than tied to actual things. Maybe because at this point, it is very hard to keep the language plain and the explanations simple. Thinking about “things” this deeply makes one feel rather existential.
Is this all just theoretical cogitations?
“No, it is very real,” Vocke says.
The silicon sphere, he patiently explains, is extremely pure. We are talking about something very tangible.
The measurement teams all assigned to this from around the world have tried to measure this tangible thing in a way that could be easily replicated. That, then, means that anyone could use the tools and the math and make their own artifact, a silicon sphere. This could be done at any point in the future and we would know for certain that we are all talking about the exact same amount of something. From now until a thousand years from now and beyond.

But this also means that they wouldn’t have to make their own artifact because they could rest assured knowing that the top measurement experts in the world had proved it could be done and had tested the results against those produced by the Kibble balances using quantum theory.
The International Avogadro Coordination (IAC) began work in 2004 as a collaboration between the members of national measurement institutes. NIST participated by confirming measurements of crystal lattices using X-ray interferometry with a natural silica crystal. It was this committee, led by Germany, including members from Russia, Italy, Belgium, Australia, Japan, the U.K. and the U.S, that later began work on redefining the mole in terms of the highly silicon-28-enriched silicon. In 2011, the preliminary results of their efforts were published in a paper in the journal Metrologia.
NIST officially became part of the IAC effort in 2012 when it was asked to participate in some of the measurement science challenges on the enriched silicon in the years that followed.
Purity = Certainty
The key, the scientists had realized, was to make silicon as pure as possible and this, in turn, would reduce the uncertainty in the measurements. In casual conversation, the word “pure” is sometimes used to describe something found in its natural state, unaltered. But in this case, pure silicon means that there are no other elements in the materials, just silicon.
This brings us back to the atomic structure of silicon itself. Silicon has three stable isotopes, known as silicon-28, silicon-29 and silicon-30. These isotopes are all silicon, but they each have slightly different weights because of differences in their atomic nuclei: They all have 14 protons (positively charged particles); this makes them silicon. However, there are different numbers of neutrons (particles with no net charge) in the nuclei, ranging from 14 for silicon-28 to 16 for silicon-30. Silicon-28 is the lightest isotope, with a mass number (protons plus neutrons) of 28, while silicon-30 is the heaviest, with a mass number of 30. This means that the molar mass of a sample of silicon could vary, depending on how much silicon-28, silicon-29 and silicon-30 it contained.
In nature, about 92 percent of silicon consists of silicon-28. The other 8 percent is made up of silicon-29 and silicon-30. If you wanted an exact molar mass of natural silicon, you would need very accurate and precise measurements of not only the amount of silicon-28, but also the amounts of silicon-29 and silicon-30 in the crystal.
Now imagine that you could change the isotopic composition of the silicon you were working with and enrich the silicon-28 so that the sample became nearly 100 percent silicon-28. Pure.
The idea here is that as the silicon becomes more and more enriched in silicon-28, the amounts of silicon-29 and silicon-30 decrease, making their contribution to the molar mass less and less significant. Silicon with 100 percent silicon-28 would have essentially no uncertainty for its molar mass.
The hard part was figuring out how to get that level of purity in the material.

Mass Speculation Becomes Mass Spectrometery
In the 1970s, Dick Deslattes and his colleagues at NIST (then called the National Bureau of Standards) used X-ray interferometry and absolute measurements of natural silicon isotopic composition using a gas of silicon hexafluoride to calculate an Avogadro constant. Their efforts with these gas sources improved the estimate of NA by nearly two orders of magnitude over previous estimates.
It was a long time before anyone could significantly improve upon the work of Deslattes and his team, in part because of their reliance on natural silicon. Even the best available instruments weren’t sophisticated enough to assess the effects of silicon contamination, given that the sample and the contaminate had nearly identical isotopic signatures.
But in the early 2000s, scientists at Germany’s national metrology institute, the Physikalisch-Technische Bundesanstalt (PTB), asked Russian scientists if they could enrich the silicon sample in silicon-28. The enrichment via a centrifugal cascade process raised the isotopic abundance of silicon-28 from 92 percent to 99.995 percent. This was a significant step forward and opened all kinds of possible experimental avenues, but it brought its own headaches as well.
How do you measure the contributions of silicon-29 and silicon-30 with sufficient accuracy when they constitute less than 0.005 percent of the atoms in a crystal? These isotopes add a slight, and almost imperceptible amount of mass to the crystal. If you could account for them, you could reduce the uncertainty of the molar mass measurement by nearly 100 times. At this stage of the investigations, the molar mass measurements were the principal factor limiting further improvements in the estimation of a more precise Avogadro constant.
X-ray diffraction-interferometry methods also improved. While mass spectrometry measures the crystal’s silicon mass, X-ray diffraction is the technique for measuring the distances between the atoms in the silicon crystal. Like all crystals, the silicon sphere consists of atoms in a regularly repeating fixed three-dimensional pattern. By combining the information about the distance between silicon atoms, the molar mass of the silicon in the crystal, and the mass and volume of the sphere, the researchers can determine the number of silicon atoms it contains. They can literally count them.
The team at PTB came up with a clever way of accurately measuring the extremely silicon-28-enriched material. The approach had been developed in the 1990s and was based on a new mass spectrometry technology that used a plasma source for ionizing the sample, to make it electrically charged. A plasma is a gas of electrically charged particles. It does not exist under normal conditions on Earth’s surface but can be generated by heating a gas within an electromagnetic field. Plasma sources for sample ionization are key to modern mass spectrometer technology.
A group of national measurement institutes then embarked on a new measurement campaign using this technology. The idea was to have experts characterize with the smallest uncertainty the isotopic composition of the enriched silicon.
The Germans cut two precise spheres from the enriched silicon, each weighing a kilogram. Although a kilogram contains more than one mole of silicon atoms, the spheres’ volumes and densities could be measured in a straightforward manner because you only need diameter to characterize a sphere’s volume. And unlike a cube or a cylinder, it is hard to knock a chip off a sphere.
This process could be replicated by anyone with the right equipment. They would just need to know exactly what the isotopic composition was for the enriched silicon to count the number of atoms accurately.
To hear Vocke tell it, this is where it begins to feel the most like a friendly science fair, albeit one that went on for more than two decades. The Germans, he says, wanted to have other national metrology institutes such as NIST participate in the research because the measurement challenges were significant. With everyone working together using slightly different approaches, any doubts about the accuracy of isotopic measurements of the enriched silicon could be removed.
“This was the big uncertainty. If we could reduce it, then we’d shrink down how we agree or don’t agree,” Vocke says.
Spheres of Certainty
The Germans had taken the silicon enriched by the Russians and used it to carefully grow an enormous purified crystal. From that, two spheres, each weighing a kilogram, were cut, shaped and polished. Then, from specific areas around the two spheres small samples were cut and then distributed to several national metrology institutes around the world for experimentation. In addition to NIST, the National Research Council Canada, the National Metrology Institute of Japan and other such organizations were asked to participate. It was really like a friendly, open competition to see who could come up with the best way to use the newest kinds of technology. The results would improve measurement for everyone.
“I guess it is sort of like: we can share the blame or share the glory, too,” Vocke says.
First, the pieces of the material had to be etched to reduce oxidation on their surfaces. Then the pieces were dissolved. And then they were analyzed by the mass spectrometer.
In looking over the Germans’ process, Rabb noticed that they were dissolving the silicon in sodium hydroxide. That seemed a possible source of trouble to him because the sodium was present in a relatively high concentration. The silicon signal would be decreased due to the huge flux of sodium ions in the ion beam. Clogging of the equipment would also be inevitable.
Rabb recalled that in past experiments with silicon he had used tetramethylammonium hydroxide (TMAH), a colorless liquid used in commercial etching and stripping mixtures. Sodium hydroxide stays in the spectrometer, but TMAH just goes away in the atmosphere—it vaporizes.
“So basically, we felt that using the TMAH wouldn’t allow a lot of material other than silicon to stay on the cones of the mass spectrometer, which can also affect the silicon signal,” Rabb says. The NIST team tried it and found that the TMAH not only was the perfect diluent for the silicon solutions but also helped to clear out anything remaining as carry-over from the previous samples.

The work took a total of three years, in part because the NIST researchers began to realize they could make the measurement process do more than they had originally thought. By 2016, they were ready to share the information with the world.
On November 16, 2018, representatives from more than 50 countries made history when they gathered in Versailles, France, to vote on redefining the SI, including the mole. The vote closed the book on this chapter of Vocke and Rabb’s work, but opened a new chapter in chemistry. The mole is no longer tied to a physical object—the carbon-12 atom. Instead, it is defined purely in terms of the Avogadro constant.
When asked where he got the idea to use TMAH in the measurement work, Rabb shrugs. While he was familiar with the materials, he thinks Vocke’s experience with the high-powered mass spectrometer was key.
“He’s downplaying it a bit,” Vocke insists, pointing back to Rabb. “But this TMAH business was so significant that all the measurements made with the sodium hydroxide diluent have been dropped from any consideration. And all measurements are now being made using TMAH, worldwide.”
To Vocke, the most amazing part was the collaborative effort made by all the different nations. “Working together!” he says with great enthusiasm. “Amazing!”

